PDF chapter test TRY NOW
1.Show the formation of Na_2O and MgO by transferring electrons.
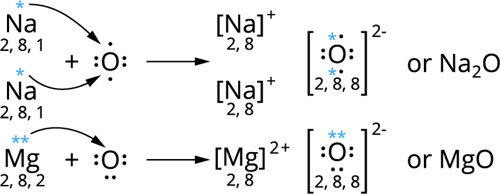
2. Why do ionic compounds have high melting points?
Positive and negative ions are present in ionic compounds. Due to oppositely charged ions , breaking this force and melting the ionic combination requires a lot of heat energy. Ionic compounds have relatively high melting points as a result of this.
