PDF chapter test TRY NOW
Isobars
Isobars are atoms with different atomic numbers but the same mass number. In other words, the nucleon count is the same, but the number of protons is different.
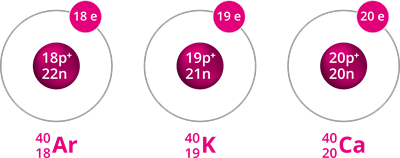
Example for isobars
Difference between isotopes and isobars:
Isotope | Isobar |
| Same atomic numbers but different mass number. | Different atomic numbers but the same mass number. |
The chemical properties are identical. | The chemical properties are different. |
| Physical properties differ from one another. | The physical properties are same. |
| The number of protons and electrons are same but the number of neutrons differs. | The number of protons, electrons and neutrons differ. |
| Examples: | Examples: |
Isotones
Isotones are atoms of different elements with different atomic numbers and mass numbers but the same number of neutrons.
Example: and
Number of neutrons in boron = 11 - 5 = 6
Number of neutrons in carbon = 12 - 6 = 6
The above pair of elements boron and carbon has the same number of neutrons but different numbers of protons, resulting in different atomic numbers.
Other examples:
1.
| (p = 6, n = 8) | |
| (p = 8, n = 8) | |
| (p = 7, n = 8) |
2.
| (p = 9, n = 10) | |
| (p = 10, n = 10) |
3.
| (p = 11, n = 12) | |
| (p = 12, n = 12) |
