PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoLet us see what an atom, molecule and element is!

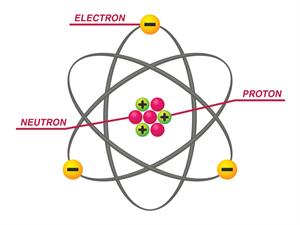
Atoms make up everything you can see, touch, smell, feel, and taste.

Atoms are the essential building blocks of all matter (including you and anyone else you will ever meet). So, let us learn a few things about these extraordinarily tiny particles if we want to understand more on this topic.

Everything you have ever seen is made up of atoms or atoms stuck together in complex configurations.

A chemical element is a material that cannot be broken down into simpler forms using traditional chemical methods.
Molecules of compounds contain atoms from two or more separate elements. For example, Ethane \(C_2H_6\) has eight atoms, two of carbon \(C\) and six of hydrogen.
An atom is the smallest unit of a chemical element. They are incredibly smaller than anything we can compare them to or imagine.