PDF chapter test TRY NOW
Activity 5.5
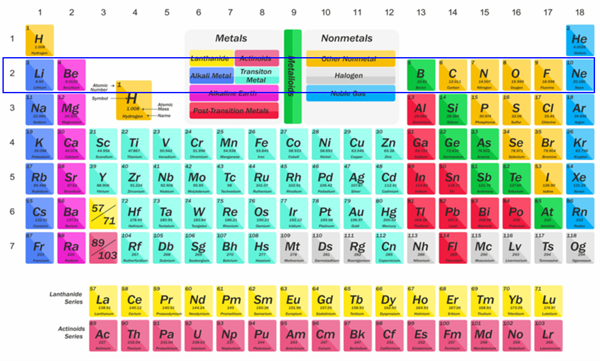
If you study the modern periodic table, you will see that the elements Li, Be, B, C, N, O, F, and Ne are present in the second period. Write down their electronic configurations.
Lithium (Li) (Atomic number: 3), electronic configuration = 2, 1
Beryllium (Be) (Atomic number: 4), electronic configuration = 2, 2
Boron (B) (Atomic number: 5), electronic configuration = 2, 3
Carbon (C) (Atomic number: 6), electronic configuration = 2, 4
Nitrogen (N) (Atomic number: 7), electronic configuration = 2, 5
Oxygen (O) (Atomic number: 8), electronic configuration = 2, 6
Fluorine (F) (Atomic number: 9), electronic configuration = 2, 7
Neon (Ne) (Atomic number: 10), electronic configuration = 2, 8
Modern periodic table group 2 elements
1. Do these elements have the same number of valence electrons?
These elements do not have the same number of valence electrons.
Lithium (Li) valence electron = 1
Beryllium (Be) valence electron = 2
Boron (B) valence electron = 3
Carbon (C) valence electrons = 4
Boron (B) valence electron = 3
Carbon (C) valence electrons = 4
Nitrogen (N) valence electron = 5
Oxygen (O) valence electron = 6
Fluorine (F) valence electron = 7
Neon (Ne) valence electrons = 0
Oxygen (O) valence electron = 6
Fluorine (F) valence electron = 7
Neon (Ne) valence electrons = 0
[Valence electrons become zero when the outermost orbital is completely filled. Thus, all inert gases have valence electrons equal to zero because their outermost orbital is completely filled. This is why they are unreactive or inert gases.]
2. Do they include the same number of shells?
Yes, they all have the same number of shells, which is two. As a result, they are all assigned to period 2.
You will find that these second-period elements do not have the same number of valence electrons, but they include the same number of shells. You also note that the number of valence shell electrons increases as the atomic number increases by one unit, moving from left to right in a period.
Or we can state that atoms of various elements with the same number of occupied shells are assigned the same period. Because the electrons in the atoms of these elements are filled in the K, L, and M shells. The elements like Na, Mg, Al, Si, P, S, Cl, and Ar belongs to the third period of the modern periodic table. Write the electronic configuration of these elements to confirm the above statement. Each period represents the filling of a new electronic shell.
