PDF chapter test TRY NOW
In the first, second, third, and fourth periods, how many elements are there?
We can describe the number of elements in these periods based on how electrons are filled into different shells. You will read about them in detail in higher classes. Recall that the maximum number of electrons accommodated in a shell depends on the formula 2n^2, where ‘n’ is the number of the given shell from the nucleus.
For example,
K Shell - 2 × (1)^2 = 2; therefore the first period has 2 elements.
L Shell - 2 × (2)^2 = 8; therefore the second period has 8 elements.
K Shell - 2 × (1)^2 = 2; therefore the first period has 2 elements.
L Shell - 2 × (2)^2 = 8; therefore the second period has 8 elements.
The third, fourth, fifth, sixth and seventh periods each have 8, 18, 18, 32 and 32 elements. The reason for this is you will study in higher classes.
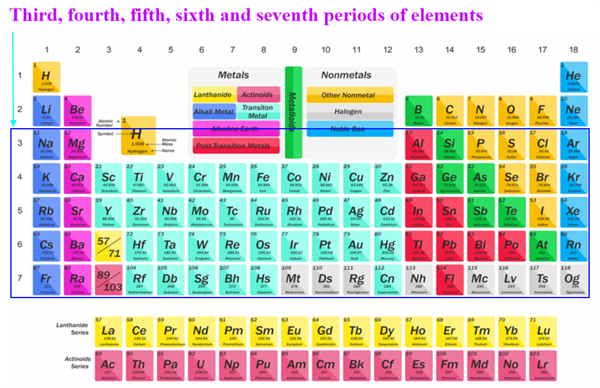
Its position in the periodic table determines an element's chemical reactivity. As you know, the number and kind of bonds formed by an element are determined by the valence electrons. Can you now explain why Mendeléev's choice of compound formulae as the basis for deciding an element's position in his table was a good one? How can this lead to elements with the same chemical properties being grouped together?
