PDF chapter test TRY NOW
Valency:
As you may know, the valency of an element is determined by the number of valence electrons present in the outermost shell of an atom.
Valence electrons in carbon
Activity 5.6
1. How do you calculate an element's valency from its electronic configuration?
After writing the electronic configuration, you will calculate the valence electrons of an element.
And valency of an element = Valence electrons of the element.
Explanation:
The combining capacity is known as valency.
Every atom is trying to complete its octet in its outermost orbit to make it stable. And atoms complete their octet by either losing electrons or gaining electrons depending upon the situation.
Atoms that have less than 4 electrons in their outermost orbit generally complete their octet by losing electrons. And atoms holding more than 4 electrons in their outermost orbit generally tend to complete their octet by gaining electrons.
The number of electrons wanted to complete the octet of an atom is called its Valency.
But atoms of element fall in first and second groups of the periodic table get stable after making duet, that means 2 electrons in their outermost orbit.
Example:
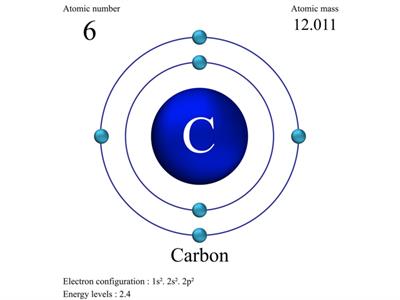
Carbon atomic number (6).
Therefore electronic configuration of carbon is 1s^2, 2s^2, 2p^2 or electronic configuration of carbon = 2, 4
Hence, number of electrons in outermost orbit in carbon atom = 4
So, valence electron of carbon = 4
Therefore, carbon requires 4 more electrons to complete its octet.
Hence, the valency of carbon = 4
So, valence electron of carbon = 4
Therefore, carbon requires 4 more electrons to complete its octet.
Hence, the valency of carbon = 4
2. What are the valency of magnesium (Atomic number 12) and Sulphur (Atomic number 16)?
Magnesium valency
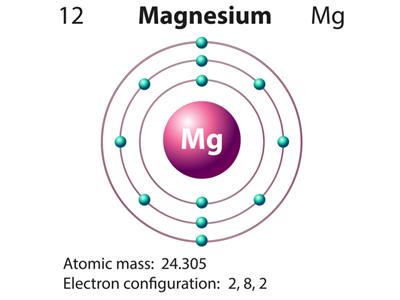
Magnesium (Atomic number 12) electronic configuration = 2, 8, 2
Magnesium valence electron = 2
Therefore, the valency of Magnesium = 2
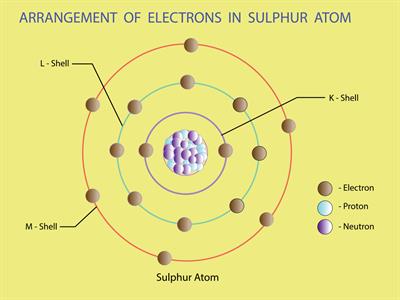
Sulphur (Atomic number 16) electronic configuration = 2, 8, 6
Sulphur valence electron = 6
Therefore, the valency of Sulphur = 2
Sulphur needs 2 more electrons to complete its octet.
3. Similarly, find the valence of the first twenty elements.
(1). Hydrogen (atomic number: 1) electronic configuration = 1
Hydrogen valence electron = 1
Therefore, the valency of Hydrogen = 1
(2). Helium (atomic number: 2) electronic configuration = 2
Helium valence electron = 0
[Because the outermost orbit is completely filled]
Therefore, the valency of Helium = 0
(3). Lithium (atomic number: 3) electronic configuration = 2, 1
Lithium valence electron = 1
Therefore, the valency of Lithium = 1
(4). Beryllium (atomic number: 4) electronic configuration = 2, 2
Beryllium valence electron = 2
Therefore, the valency of Beryllium = 2
(5). Boron (atomic number: 5) electronic configuration = 2, 3
Boron valence electron = 3
Therefore, the valency of Boron = 3
(6). Carbon (atomic number: 6) electronic configuration = 2, 4
Carbon valence electron = 4
Therefore, the valency of Carbon = 4
(7). Nitrogen (atomic number: 7) electronic configuration = 2, 5
Nitrogen valence electron = 5
Therefore, the valency of Nitrogen = 5 or 3 (Variable valency)
[Because nitrogen is an electronegative element, it can receive three electrons, resulting in a valency of 5 or 3.]
(8). Oxygen (atomic number: 8) electronic configuration = 2, 6
Oxygen valence electron = 6
[Oxygen is an electronegative element, so it completes its octet by gaining electrons because it needs only two electrons to complete its octet.]
Therefore, the valency of Oxygen = 2
(9). Fluorine (atomic number: 9) electronic configuration = 2, 7
Fluorine valence electron = 7
[Fluorine is an electronegative element, so it completes its octet by gaining electrons because it has one less electron to complete its octet.]
Therefore, the valency of Fluorine = 1
(10). Neon (atomic number: 10) electronic configuration = 2, 8
Neon valence electron = 0
[As neon has completely filled the outermost orbit, therefore its valence electron will become equal to zero.]
Hence, the valency of Neon = 0
(11). Sodium (atomic number: 11) electronic configuration = 2, 8, 1
Sodium valence electron = 1
Therefore, the valency of Sodium = 1
(12). Magnesium (atomic number: 12) electronic configuration = 2, 8, 2
Magnesium valence electron = 2
Therefore, the valency of Magnesium = 2
(13). Aluminium (atomic number: 13) electronic configuration = 2, 8, 3
Aluminium valence electron = 3
Therefore, the valency of Aluminium = 3
(14). Silicon (atomic number: 14) electronic configuration = 2, 8, 4
Silicon valence electron = 4
Therefore, the valency of Silicon = 4
(15). Phosphorous atomic number: 15) electronic configuration = 2, 8, 5
Phosphorous valence electron = 5
[Because phosphorous can gain three electrons to complete its octet.]
Therefore, the valency of Phosphorous = 5 or 3 (Variable valency)
(16). Sulphur atomic number: 16) electronic configuration = 2, 8, 6
Phosphorous valence electron = 6
Because Sulphur completes its octet by accepting two electrons,
Therefore, the valency of Sulphur = 2
(17). Chlorine (atomic number: 17) electronic configuration = 2, 8, 7
Chlorine valence electron = 7
[Chlorine completes its octet by gaining one electron because it is an electronegative element.]
Therefore, the valency of Chlorine = 1
(18). Argon (atomic number: 18) electronic configuration = 2, 8, 8
The number of electrons in the outermost orbit is 8.
[It has a completely filled outermost orbit, whereas elements filled outermost orbits have zero valence electrons.]
Hence, Argon valence electron = 0
Therefore, the valency of Argon = 0
(19). Potassium (atomic number: 19) electronic configuration = 2, 8, 8, 1
Potassium valence electron = 1
Therefore, the valency of Potassium = 1
(20). Calcium (atomic number: 20) electronic configuration = 2, 8, 8, 2
Calcium valence electron = 2
Therefore, the valency of Calcium = 2
4. How does the valency of a period change from left to right?
When going from left to right in a period, the valency increases up to four and then decreases.
5. How does the valency change as you move down the group?
The valency of an element does not change as it moves down in a group.
