
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
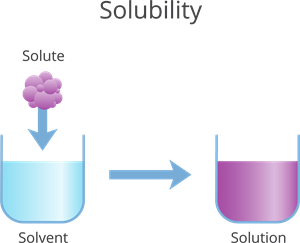
Book Free DemoA solution is a homogeneous mixture of two or more substances.
- A solute is a part of a solution that is present in a smaller quantity by weight.
- A solvent is a variable that is present in a greater quantity by weight.
The solvent is the substance that does not dissolve. A solvent is a variable that is present in a greater quantity by weight.

Solvent
The solute is the material that dissolves. A solute is a part of a solution that is present in a smaller quantity by weight.

Solute
If we add the solvent and solute, we get the solution.
Solution = Solvent + Solute.

Solution formation
The solute is distributed uniformly throughout the solvent, resulting in a homogeneous mixture. The solvent serves as a dissolving medium in a solution.
The process of uniform distribution of solute into the solvent is called dissolution.
All solutions are mixtures, but all mixtures are not solutions. Why?
A solution is a homogeneous mixture that has the same or similar properties throughout the mixture. Consider the example of saltwater. This is also referred to as a "homogeneous mixture."
A mixture is not uniform throughout and so not considered as a solution. Consider the example of sand in water. This is also referred to as a "heterogeneous mixture."
A solution must have at least two components (a solute and a solvent). Binary solutions are those that consist of one solute and one solvent (two components).

Formation of copper sulphate solution
For example, when copper sulphate crystals are added to water, they dissolve and form a copper sulphate solution. Here the solution is made up of two parts namely: one solute (copper sulphate) and one solvent (water). So, it is a binary solution.

Adding mixture of salt and sugar to water
A solution may also have more than two components. When salt and sugar are added to water, they dissolve and form a solution. In this case, two solutes are dissolved in a single solvent.
Ternary solutions are solutions that have three components.

Coffee is the best example of ternary solutions as it contains three constituents such as milk, coffee powder and sugar.