
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoFour blocks of elements:
- We know that the electrons in an atom are arranged in shells around the nucleus.
- Each shell comprises one or more subshells in which the electrons are shared in a particular manner. These subshells are named s, p, d, and f.
- Based on the order of electrons in subshells, the periodic table elements are divided into four blocks: s, p, d and f.
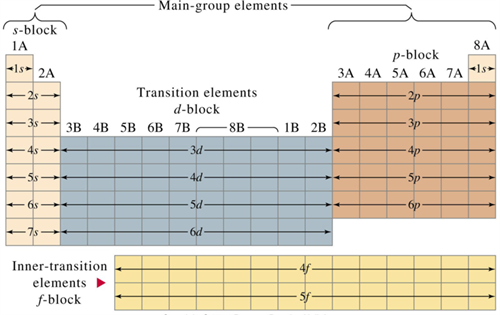
s-Block Elements:
1. Alkali metals:
- It contains groups 1 (alkali metals) and 2 (alkaline earth metals) elements.
- They are also named as representative elements.
- Except for hydrogen, the elements in group 1 are metals.
- When they react with water, they form solutions that turn red litmus paper in to blue.
- These solutions are classified as highly alkaline or basic. As a result, they are known as alkali metals.
2. Alkaline earth metals:
- The group 2 elements are metals.
- They react with oxygen to form oxides, formerly known as 'earth,' and these oxides contain alkaline solutions when dissolved in water.
- As a result, these elements are known as alkaline earth metals.
p-Block Elements:
- These elements are found in periodic table groups (13-18).
- In addition to noble gases, they include the boron, carbon, nitrogen, oxygen, and fluorine families (except helium).
- They are also named as representative elements.
- The p-block is home to the widest variety of elements and is the only block that includes all three types of elements: metals, non-metals, and metalloids.
d-Block Elements:
- It contains elements from groups (3 to 12).
- They are located in the middle of the periodic table.
- These block elements' properties are intermediate to that of s and p block elements. Hence, they are called transition elements.
f-Block Elements:
- It contains 14 elements named Lanthanoides after (Lanthanum) La (57) and 14 elements called Actinoides after (Actinium) Ac (89).
- They are fixed at the bottom of the periodic table.
- They are also named as inner transition elements.
Reference:
https://upload.wikimedia.org/wikipedia/commons/b/b6/PTable_structure.png