
PUMPA - SMART LEARNING
எங்கள் ஆசிரியர்களுடன் 1-ஆன்-1 ஆலோசனை நேரத்தைப் பெறுங்கள். டாப்பர் ஆவதற்கு நாங்கள் பயிற்சி அளிப்போம்
Book Free DemoFeatures of Coordinate Covalent Compounds:
The compounds, including coordinate covalent bonds, are named coordinate compounds.
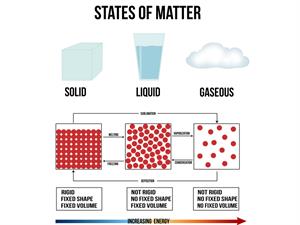
Physical state:
These compounds can be found as gases, liquids or solids state.
Electrical conductivity:
Coordinate compounds, like covalent compounds, lack charged particles (ions), making them poor conductors of electricity.

Electrical conductivity
Melting point: These compounds possess higher melting and boiling points than pure covalent compounds but are lower than pure ionic compounds.

Covalent compound melting
Solubility:
Benzene, \(CCl_4\) and other non-polar solvents make these compounds soluble. They are insoluble in water and polar solvents.

Covalent compound solubility
Reactions: